Density of water gm34/21/2024  Since volume is in the denominator, increasing the volume decreases the density. As you heat something up, the volume usually increases because the faster moving molecules are further apart. Why does the density of water change with temperature?ĭensity changes with temperature because volume changes with temperature. Ice is less dense than liquid water which is why your ice cubes float in your glass. The density of water is roughly 1 gram per milliliter but, this changes with temperature or if there are substances dissolved in it. Impure water density increases with respect to impurity. The density of pure water varies with temperature and attains its maximum value at a temperature of 4^0C. What are the factors that affect the density of water?Īnswer: There are two main factors which affect the density of water, namely, Temperature and Purity. The specific volume v is given by: v = 1 /r. There is a related state variable called the specific volume which is the reciprocal of the density r. Gas density is defined to be the mass of gas divided by the volume confining the gas. It means the amount of three-dimensional space a closed figure can occupy is measured by its volume. Volume is a three-dimensional quantity that is used to measure the capacity of a solid shape. A block of Styrofoam is less dense than a brick. For example, a block of the heavier element lead (Pb) will be denser than the softer, lighter element gold (Au). 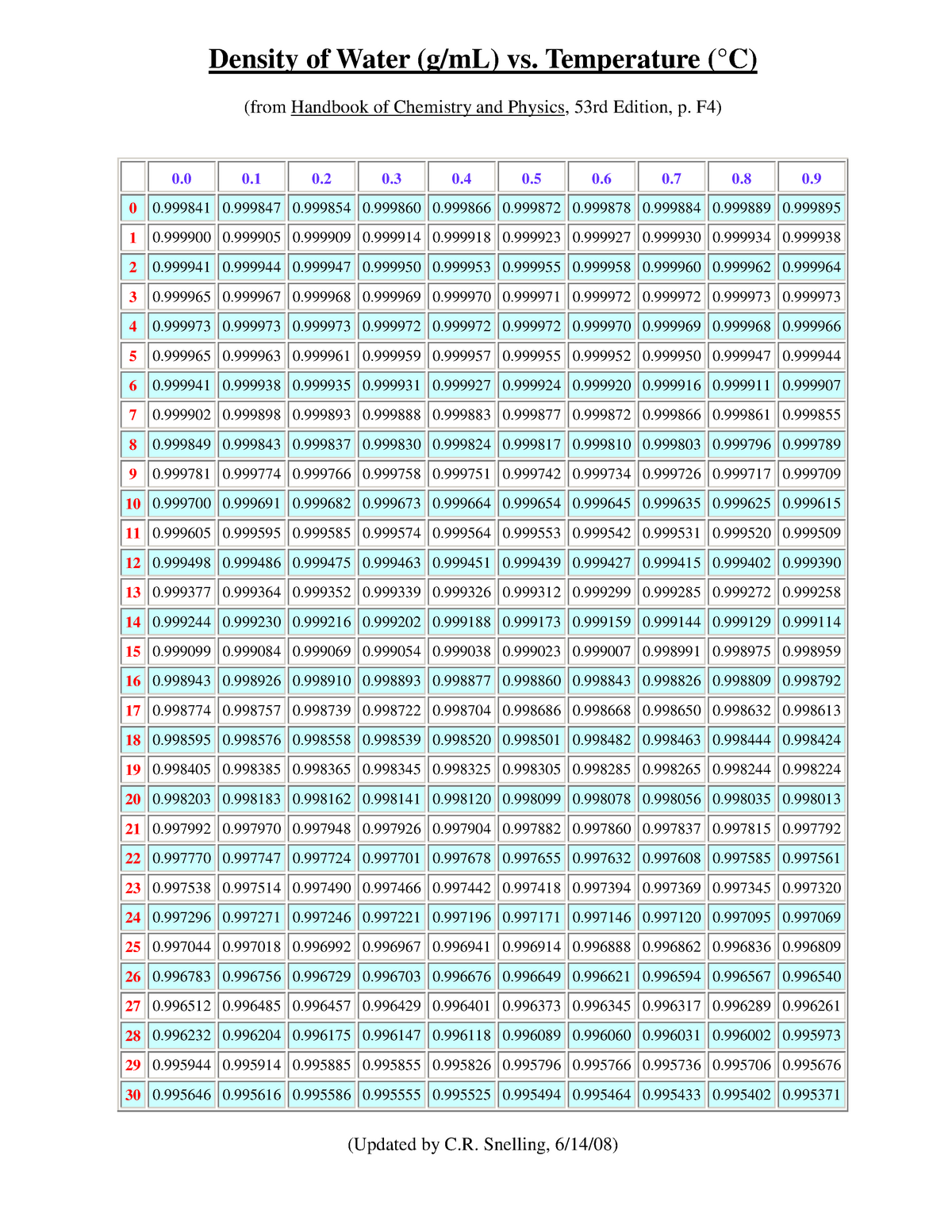
What is density with example?ĭensity is the measure of how much “stuff” is in a given amount of space. Since the density of a liquid varies as its temperature changes, the scale is adjusted to a certain temperature, usually about 15 degrees C., at which determinations must be made. Thus, the density of water is a maximum at 4 ☌. 
Cluster formation is the bigger effect, so the density starts to decrease. The molecules are still slowing down and coming closer together, but the formation of clusters makes the molecules be further apart. 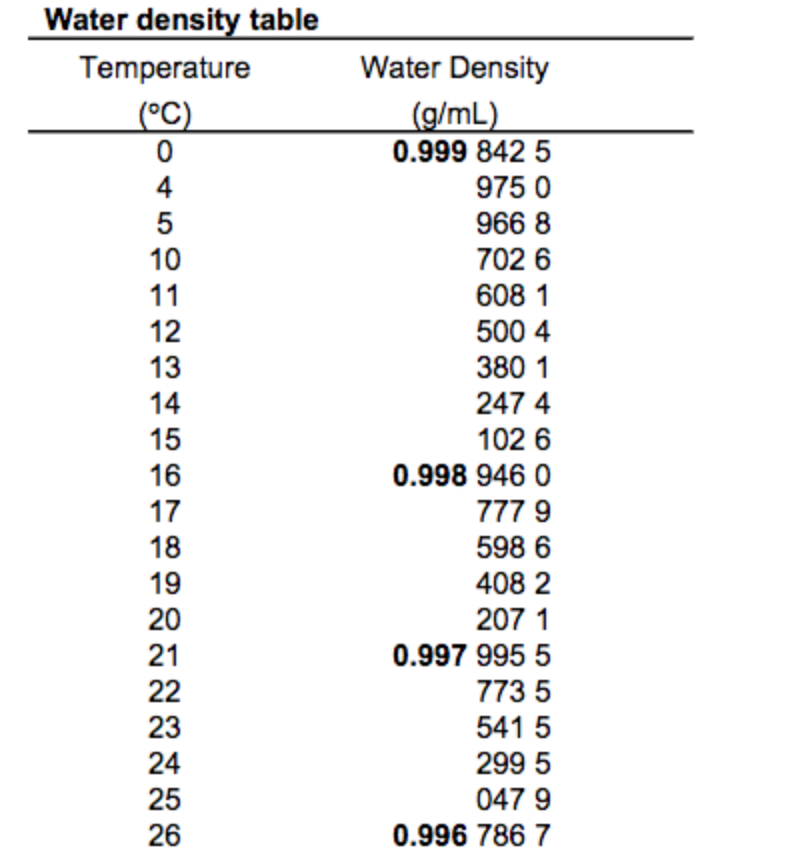
What is the density of 100% pure water?ġ.000 g/mL at 3.98 ☌ (lit.) Why is the density of water highest at 4?Īt 4 ☌, the clusters start forming. The units generally used for solids and liquids are g/mL, for gases g/L. The density of a material may be defined as mass per unit volume. What is the unit for density of a liquid? What is the unit for density?ĭensity has the units of mass divided by volume such as grams per centimeters cube (g/cm3) or kilograms per liter (kg/l). Kg is the SI unit of mass, m3 is the unit of volume. The substance having a density more than water will sink in it and a substance having a density less than water will float on it. Upon freezing, the ice density decreases by about 9%. The density of ice is less than liquid water, so it floats. The maximum density of water is at 4☌ as there are two opposite effects that are in balance. In imperial measurements, it’s about 62.4 pounds per cubic foot at 77 degrees Fahrenheit. In the metric system, which is more widely used around the world and in cooking, the unit weight of water is approximately 1 gram per cubic centimeter at 25 degrees Celsius, according to the National Library of Medicine. So, 1g/1cm3 = 1 g/cm3, giving water its easy-to-remember density. Density is mass divided by volume (ρ=m/v), and water was used as the basis for establishing the metric unit of mass, which means a cubic centimeter (1cm3) of water weighs one gram (1g). It’s no coincidence that water has a density of 1. Water density changes with temperature and salinity. Water is densest at 3.98☌ and is least dense at 0☌ (freezing point). 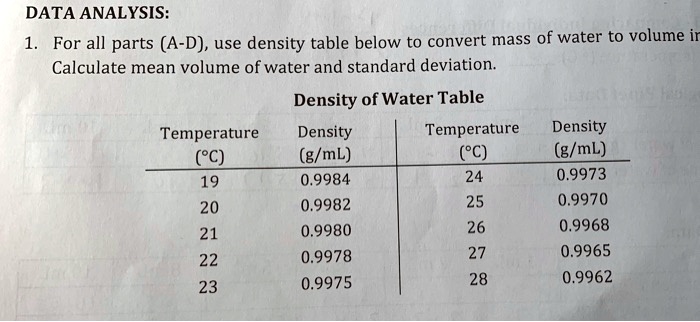
Density is measured as mass (g) per unit of volume (cm³). Density is commonly expressed in units of grams per cubic centimetre. The formula for density is d = M/V, where d is density, M is mass, and V is volume. How do you calculate density?ĭensity, mass of a unit volume of a material substance. Pure water has a density of 1g/cm 3 or 1000kg/m 3. The density is defined as the mass per unit volume ratio. Water has a density of 997 kg/m 3 at 25 degrees Celsius. The density of a substance is the same regardless of the size of the sample. The density of water is 1 gram per cubic centimeter. Just like a solid, the density of a liquid equals the mass of the liquid divided by its volume D = m/v. 24 Is density the same as specific gravity? What is the formula for density of water? 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
